By: The Trek News Desk
G. Ranganathan, the 75-year-old owner of a pharmaceutical company, was arrested early Thursday morning at his apartment in Kodambakkam, Chennai. The arrest follows a growing investigation into the deaths of several children in Madhya Pradesh and Rajasthan who allegedly consumed a contaminated batch of Coldrif cough syrup manufactured by his firm.
The incident, which surfaced after multiple fatalities in Chhindwara district of Madhya Pradesh, has brought serious concerns about the quality and safety of pharmaceutical products being distributed across the country. Authorities believe that the Coldrif syrup, found to be laced with toxic substances, directly contributed to the children’s deaths.
Arrest and Ongoing Investigation
A seven-member team from the Madhya Pradesh Police, led by Deputy Superintendent of Police Jitendra Jaat, tracked down and arrested Ranganathan from his residence around 12:30 am. Following the arrest, he was taken to Kanchipuram district for further questioning, where his company, Sresan Pharmaceutical Manufacturer, is based.
Initial sample testing of the syrup raised serious red flags about possible toxic adulteration, prompting a broader investigation into the company’s production standards and compliance with drug safety protocols.
Deadly Contaminant Found in Coldrif Syrup
State authorities in Madhya Pradesh and other regions have now banned the sale and distribution of Coldrif syrup after lab tests confirmed the presence of a dangerous industrial chemical. A forensic analysis conducted by the Government Analyst at the Drug Testing Laboratory in Chennai found that the syrup contained 48.6% Diethylene Glycol by volume, a highly toxic compound typically used in industrial products like antifreeze.
The official lab report labelled the syrup as “Not of Standard Quality” and stated:
“The sample was found to be adulterated, as it contains Diethylene Glycol (48.6% w/v), a poisonous substance that renders the product hazardous to human health.”
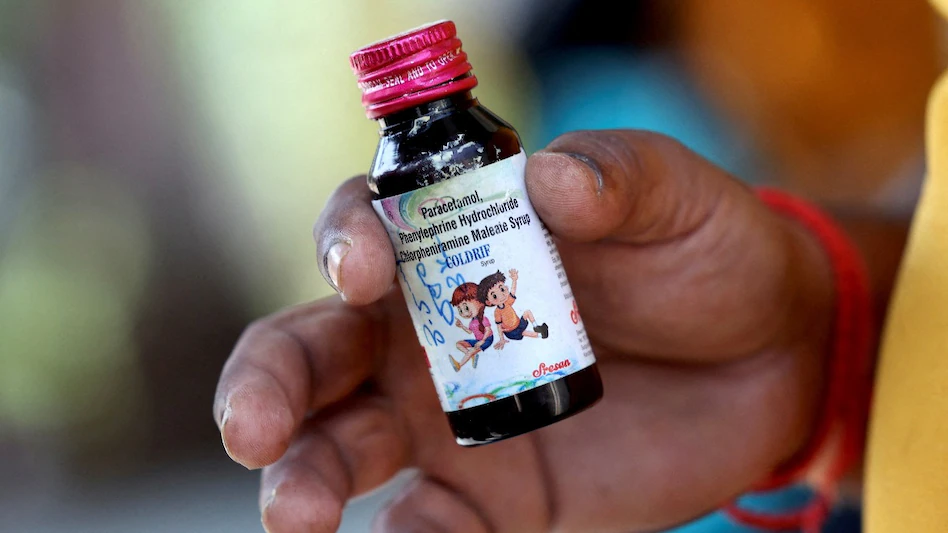
Impact on Public Health
Diethylene Glycol is not meant for human consumption and is known to cause severe damage to internal organs, particularly in children. Health experts warn that even small doses of the compound can lead to kidney failure, neurological damage, and death.
In this case, the Coldrif syrup was found to have dangerously high levels of the chemical, raising urgent alarms across the healthcare sector.
National Scrutiny and Policy Questions
The tragedy has triggered nationwide outrage and a wave of inspections across pharmaceutical manufacturing units. Questions are now being raised about the effectiveness of India’s drug regulation system, with many demanding stricter oversight and faster response mechanisms.
The Ministry of Health has taken serious note of the case and is expected to issue a series of directives aimed at tightening quality control across the pharmaceutical industry. Officials have also instructed other state governments to halt the sale of any Coldrif stock and submit samples for immediate testing.
Looking Ahead: Regulatory Overhaul Needed
Ranganathan’s arrest marks a pivotal moment in what could become a landmark case for pharmaceutical regulation in India. With the investigation still ongoing, more arrests and factory inspections are expected in the coming days.
This incident has once again spotlighted the urgent need for robust drug monitoring systems and strict accountability for manufacturers. As grieving families seek justice, the nation watches closely, hoping that this tragedy will lead to long-overdue reforms in the way medicines are produced, tested, and sold in India.
Source: News Agencies



